Author | Erin Hall, ICLAC member
Informing the research community about instances of cell line misidentification and cross-contamination is one of the core missions of ICLAC, but our mission doesn’t just focus on human cell lines. In this case study, we are focusing on RGC-5, a non-human cell line originally thought to be derived from rat retinal ganglion cells.
This case study is available for download | click here for PDF
RGC-5 is also the subject of a news feature | see The Scientist website
RGC-5: proposed to be the first transformed cell line from rat retinal ganglion cells
The ultimate cause of vision loss in patients with glaucoma is thought to be progressive loss of retinal ganglion cells (RGCs) and their axons, and it is the second leading cause of blindness worldwide (1).
In 2001, Krishnamoorthy et al. published a paper on the first virally transformed retinal ganglion cell culture isolated from a postnatal day 1 rat (2). The authors made a point to say that, at that time, there were no permanent retinal ganglion cell lines, only primary cell cultures. Primary cultures had limited uses in the research lab due to a rapid decrease in viable RGCs over time in culture. The new cell line, named RGC-5, was considered a useful model for understanding RGC biology, including mechanisms of ganglion cell death in glaucoma (2).
(1) Citation in PubMed (2) Citation in PubMed
RGC-5 reported to be of mouse origin
In September 2009, more than eight years after the RGC-5 source paper was published, Van Bergen et al. published a paper (3) disputing the origin of the RGC-5 cell line.
Back in February and June of 2007, the authors obtained samples of RGC-5 (passage 19) directly from the source of the RGC-5 cell line (University of North Texas Health Science Center (UNTHSC)). Van Bergen et al. noted that recent publications had reported increased resistance to glutamate in the RGC-5 cell line, a characteristic that contradicted the original reports on the RGC-5 cell line. The authors tested a region of mitochondrial DNA (mtDNA) that showed sequence variation between mouse and rat to identify the species of the cell line. They also performed glutamate sensitivity assays, immunohistochemistry (IHC) and other tests to fully re-characterize the RGC-5 cell line.
Van Bergen et al. found that the RGC-5 cell line that they received directly from UNTHSC contained mouse DNA, not rat as expected (a conclusion based on comparing the mouse and rat Thy-1 gene and mtDNA sequence). They also found that the glutamate sensitivity test (at 5mM glutamate) showed a 95% survival rate. This survival rate was quite unlike earlier studies by UNTHSC showing only a 35%-50% survival rate (4,2). In addition, the RGC-5 cells lacked retinal ganglion cell-specific protein markers when assessed by immunoblotting and IHC.
In summary, the RGC-5 cell line as tested by Van Bergen et al. had lost most of the RGC characteristics described by Krishnamoorthy et al. in the 2001 paper, and could not be identified as a rat retinal ganglion cell line.
(3) Citation in PubMed (4) Citation in PubMed
RGC-5 is now known to correspond to mouse photoreceptor cell line 661W
Thomas Yorio’s group, at UNTHSC, worked with the originators to analyze low passage stocks of RGC-5 (passage 2 and above). Their work, published in August 2013, confirmed the species of RGC-5 as mouse. The authors also concluded that the contaminant for RGC-5 was 661W, a mouse photoreceptor cell line developed by Al-Ubaidi et al. in the early 90’s (5,6).
It was noted that 661W cells had been present in the Krishnamoorthy laboratory. Yorio’s group found many similarities between RGC-5 and 661W cells, including the presence of SV40 large T-antigen, which was expected for 661W but not for RGC-5; the latter cells having been transformed using the ΨE1A virus. Yorio’s group employed other methods such as G-banding karyotyping and DNA typing to conclude that RGC-5 is misidentified and consists of 661W mouse cells, not rat retinal ganglion cells, in agreement with Van Bergen et al (3).
(5) Citation in PubMed (6) Article in PubMed Central
A journal’s response to RGC-5: Molecular Vision now requires authentication testing
In August 2013, the editors of journal Molecular Vision published a commentary on RGC-5. The commentary established an updated policy for use of cell lines by researchers submitting an article to Molecular Vision (7). The editors reviewed work from Yorio’s group and others to conclude, “it is likely that from [passage] 2 on, all RGC-5 clones were instead 661W cells”. Therefore, Molecular Vision has revised their policy on the use of RGC-5 to read:
New manuscripts containing data derived from RGC-5 cells will be editorially rejected without review.
In addition, the editors set out compulsory requirements for data demonstrating that any cells used in a manuscript submitted for publication in the journal exhibit the correct phenotype and genotype, including species of origin. The authentication information for each cell line must be included in the Methods section of the manuscript, as part of the freely-available article.
In December 2013, the original 2001 RGC-5 source article (2) was retracted at the request of the authors (8) and the Editor-in-Chief of the journal Brain Research (9).
(7) Journal Website (8) Journal Website (9) Journal Website
Is RGC-5 still being used as a rat retinal ganglion cell line?
Between 2001 and the end of 2013 (when the source article was retracted), there were 236 publications using “RGC-5” cells. Of those 236 articles, 130 (55%) were published AFTER the release of the 2009 paper by Van Bergen et al. cautioning researchers on use of RGC-5 as a rat retinal ganglion cell line.
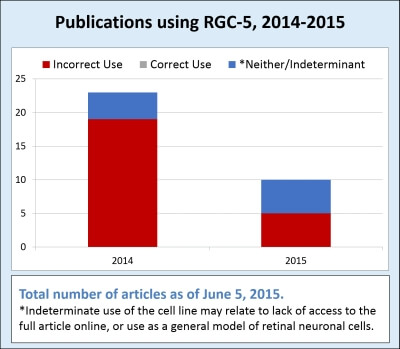
Following the retraction, there have been 33 articles published using RGC-5 cells (1 January 2014 through 5 June 2015). Of those 33 articles, 19 (58%) published experimental data using RGC-5 as a model for retinal ganglion cells. Five additional papers published RGC-5 data, making a point to say that RGC-5 is a mouse “retinal ganglion” cell line. This brings the total number of research articles that incorrectly cited the species of origin and/or cell type to 24 articles (73%) during 2014-2015.
Several of the articles mentioned the “recent” controversy over the identity of the RGC-5 cell line, but kept the data using this cell line in the paper as a model for “retinal ganglion” cells instead of photoreceptor cells.
Because of the scarcity of immortalized retinal ganglion cell lines, many of these research articles utilize RGC-5 as the sole model for experimental work. Their results are meant to add understanding to the causes and treatments for diseases of the eye. Continuing to use RGC-5 as a model for retinal ganglion cells only pushes research further off course as it tries to reveal the cause or find a cure for conditions such as glaucoma and/or diabetic retinopathy.